Control of spin states of spin-crossover compounds
 分类:research
分类:research
 作者:
作者:
 发布时间:2019-05-13
发布时间:2019-05-13
Spin crossover (SCO) refers to the spin-state interconversion of transition-metal ions located in suitable coordination environments. This conversion usually leads to or is accompanied with changes in the optical, magnetic, conductive, dielectric, mechanical, structural properties, and so on. The convertible spin states and subsequent different chemical and physical properties make SCO compounds switchable materials that can be applied in various fields.
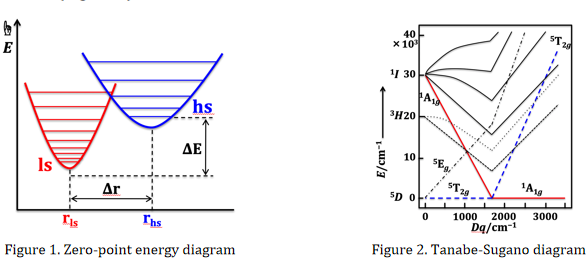
The occurrence of SCO is frequently observed in Fe(II), Fe(III) and Co(II) compounds, and occasionally observed in Cr(II), Mn(II), Mn(III), Co(III) and Ni(II) ones. When the difference of zero-point energy (ΔE) between the high-spin (hs, seldom intermediate spin state) and low-spin states is close to kBT (~ 200 cm–1 at 295 K, kB is the Boltzmann constant), the compound may undergo SCO (Figure 1).
To synthesize SCO compound, the design of ligand playing moderate crystal-field strength is very important. Taken Fe(II) ion in octahedral coordination geometry as an example, when the crystal field splitting ΔO (10 Dq, cm–1) are 11500–12500 and 19000–21000 for high-spin and low-spin species, respectively, the Fe(II) ion is most likely SCO active (Figure 2).
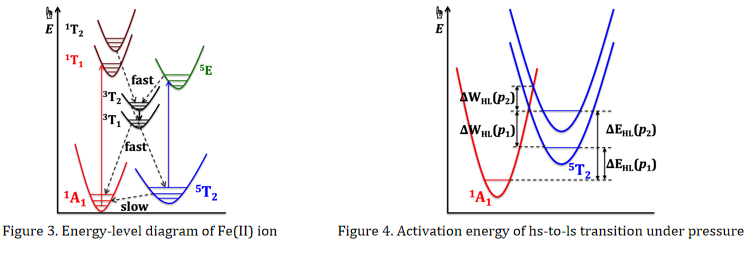
For a SCO compound, the hs ⇌ ls switching can not only be triggered by temperature changing (critical temperature T = ΔH/ΔS), but also be controlled by light irradiation (Figure 3) and pressure (Figure 4). On the other hand, some chemical processes can significantly affect the SCO behaviors, for instances desorption-sorption of guest solvent molecules, polymorphic transformation, etc.
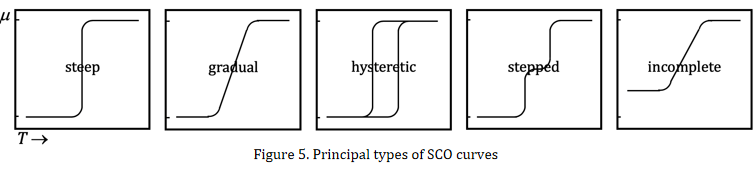
SCO process can be monitored by some physical technologies, of which magnetic measurement is most commonly used. If consider a spin-only Fe(II) ion, its hs state has four unpaired electrons and thus the magnetic moment is μ = 4.90 μB, whereas the ls state has zero unpaired electron and μ = 0. So, the magnetic moment versus temperature plot of SCO compound usually features an S-shaped curve. In fact, SCO behavior is complicated, and various types of μ versus T plots have been documented (Figure 5).
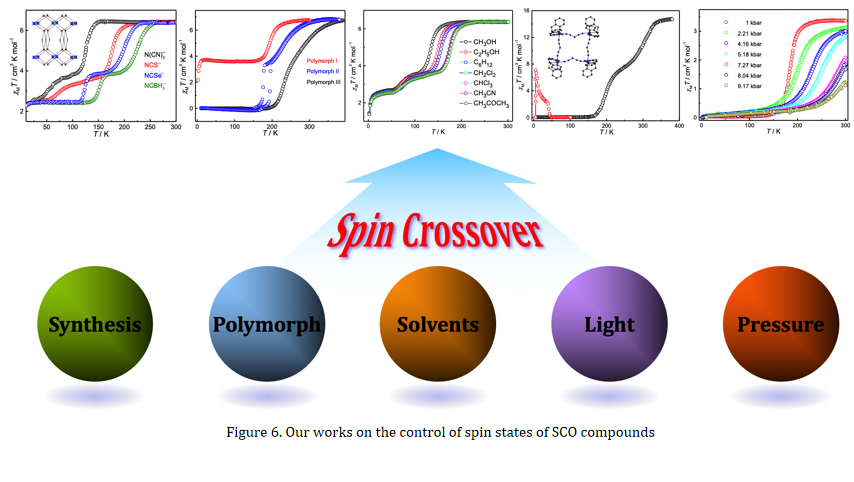
Our research interest in this field focuses on the control of spin states of SCO compounds by both chemical and physical approaches. In the past few years, we have prepared several series of Fe(II) SCO compounds based on rationally designed synthetic strategy, and have successfully controlled their spin states and tuned their SCO behaviors (Figure 6).